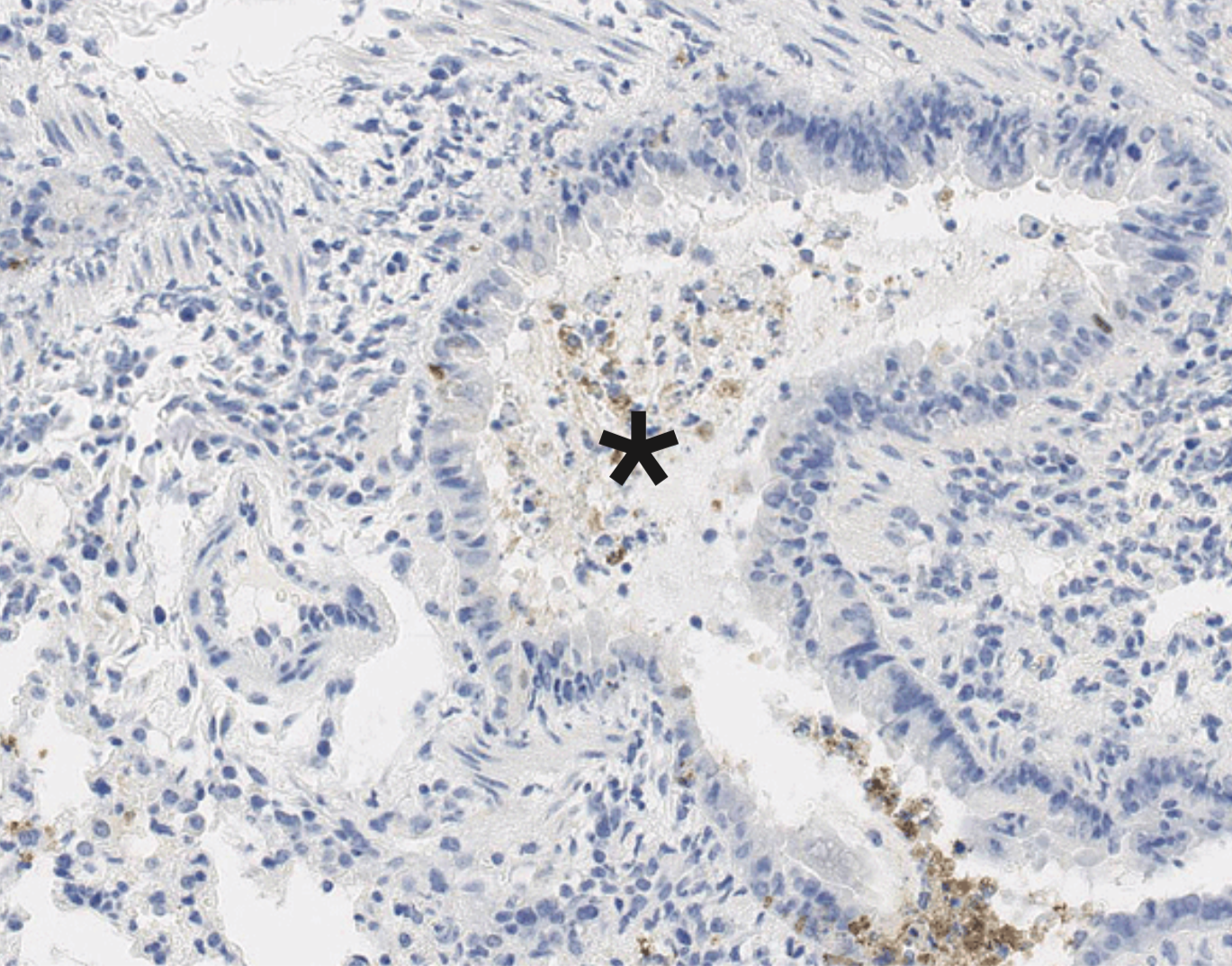
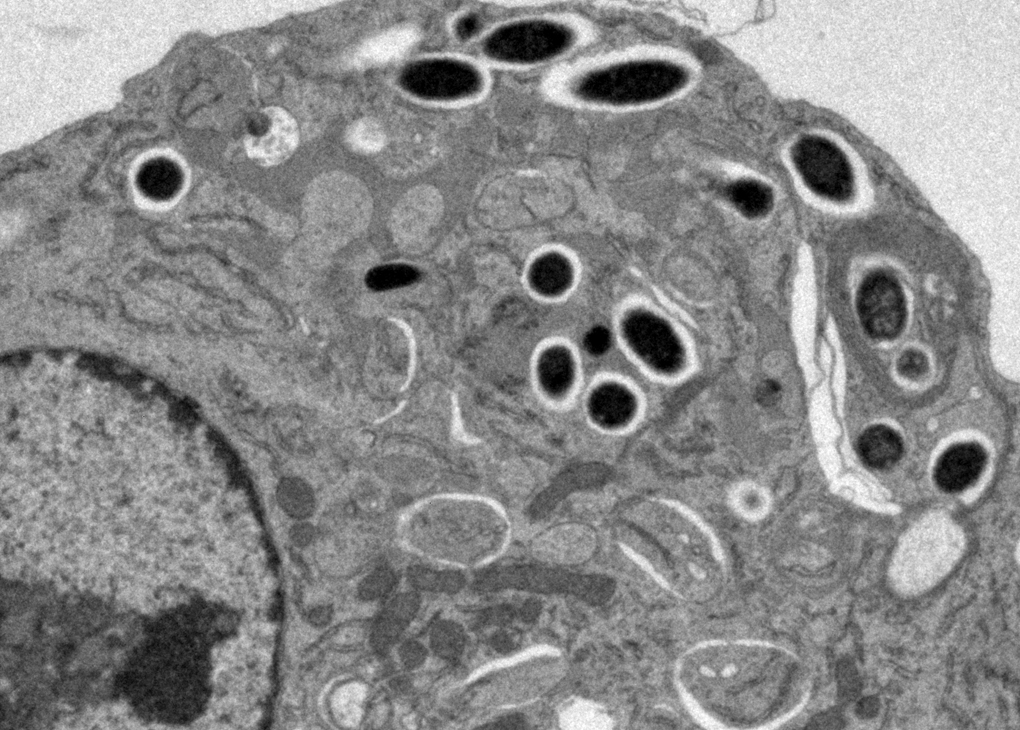
OUR RESEARCH GOALS
Our laboratory is interested in understanding how mucosal immune responses are coordinated to maintain homeostasis and respond to microbial infection, barrier disruption, or alterations in commensal microbial diversity — with an emphasis on how these molecular decisions are balanced within the context of host fitness and organ physiology. Our studies are geared toward uncovering pathways with the potential for therapeutic manipulation, specifically focusing on the signals that drive pro- and anti-inflammatory immune responses within each setting. Deciphering the molecular inputs that drive these opposing fates, and the subsequent cellular and molecular signals that immune cells employ, has broader applications in the treatment of infectious disease, cancer and autoimmune disorders.
IMMUNE HOMEOSTASIS
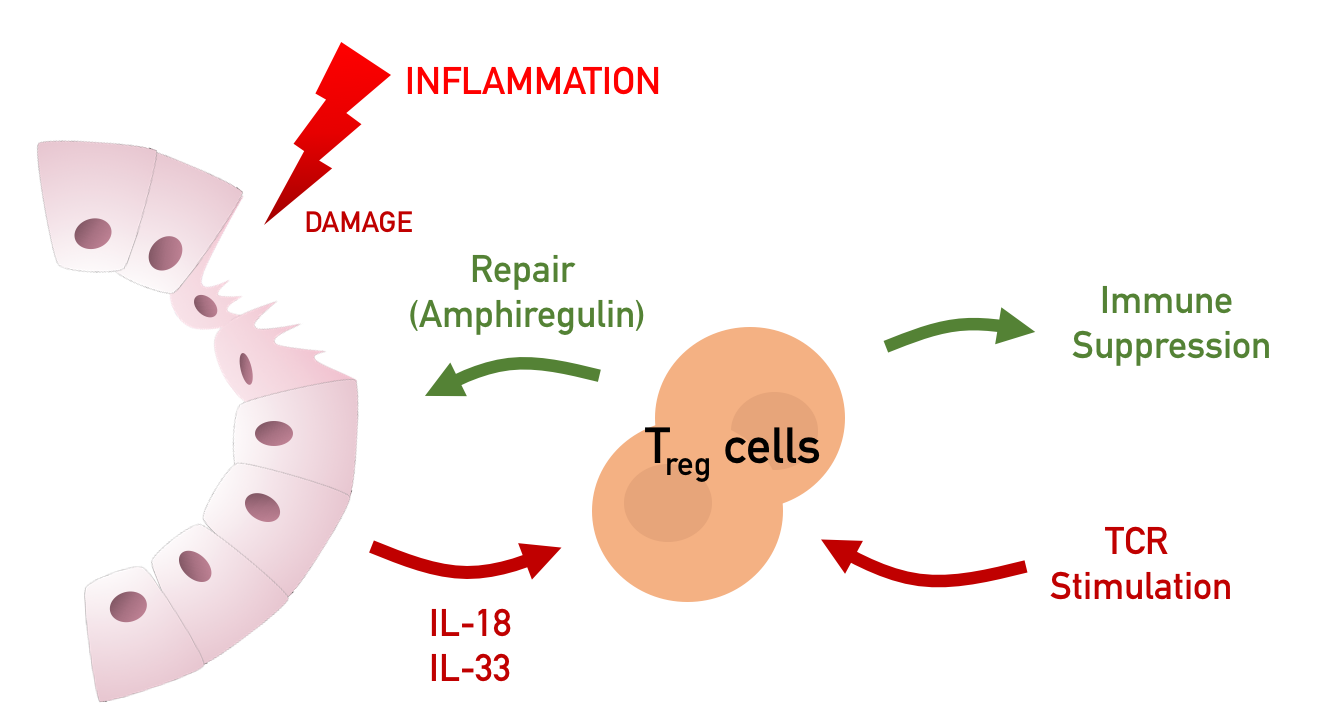
Mounting an immune response is an energetically costly endeavor that can greatly impact host fitness. In order to neutralize invading pathogens and limit collateral damage to surrounding tissues, the immune system carefully balances pro-and anti-inflammatory responses to ensure that the magnitude of a response is proportional to the pathogenic threat encountered. A cost-benefit relationship — between the collateral damage associated with an inflammatory response and the benefit of inflammation in protecting the host from infection — imparts the selective pressure for maintaining homeostasis.
MUCOSAL IMMUNITY
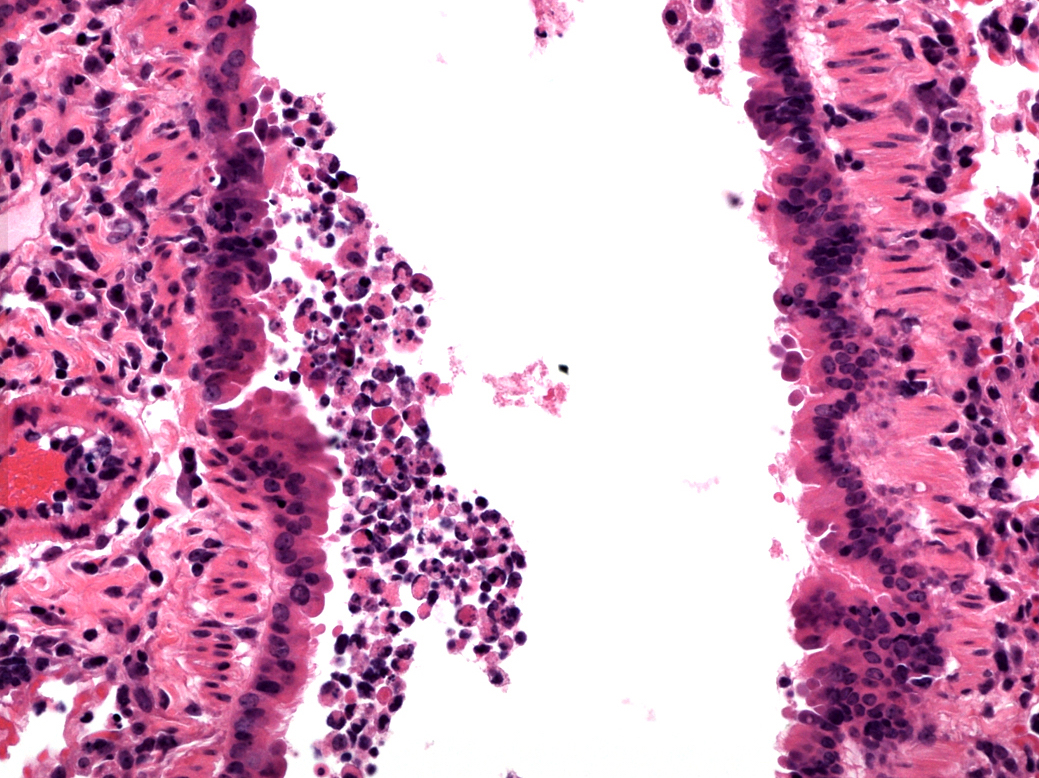
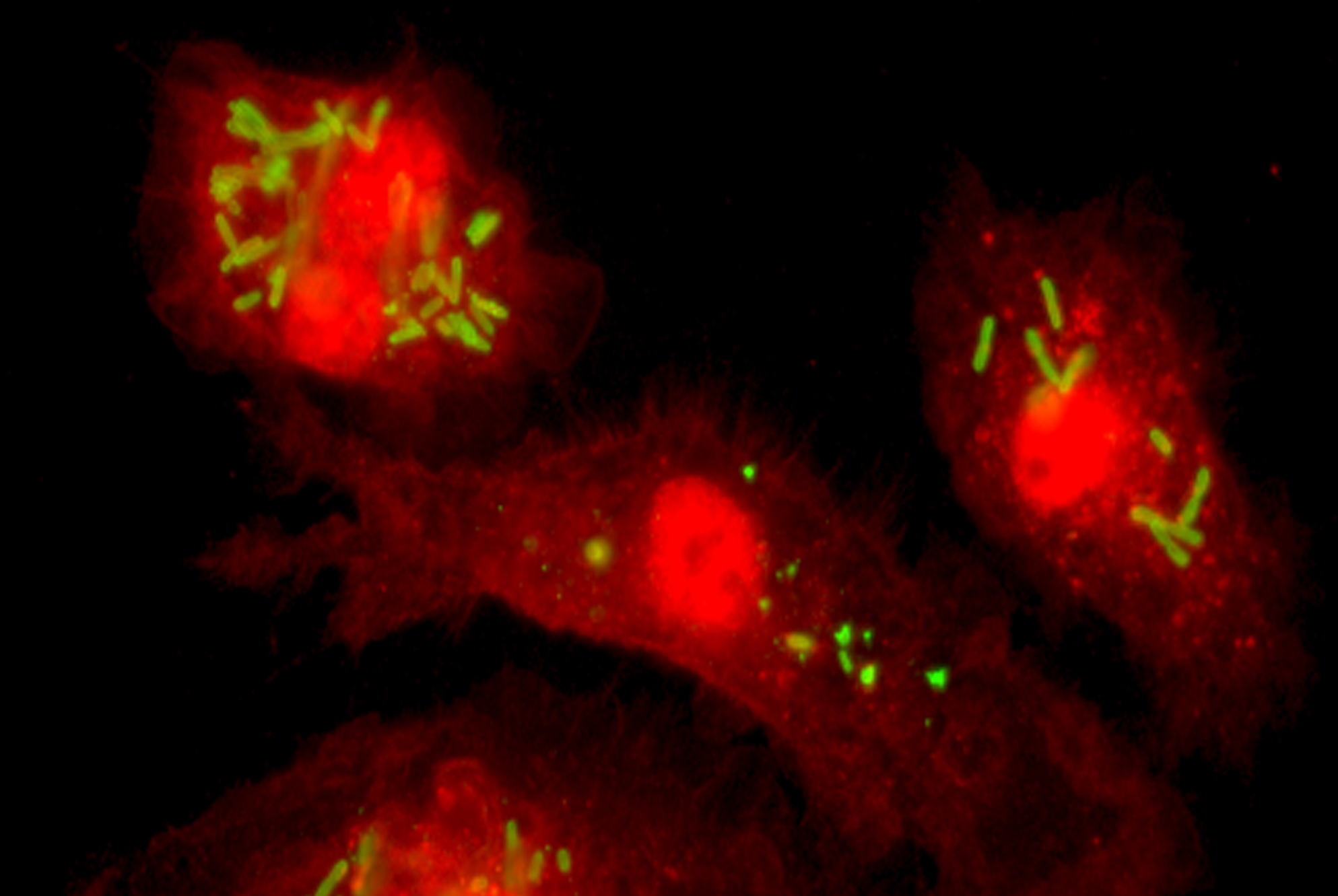
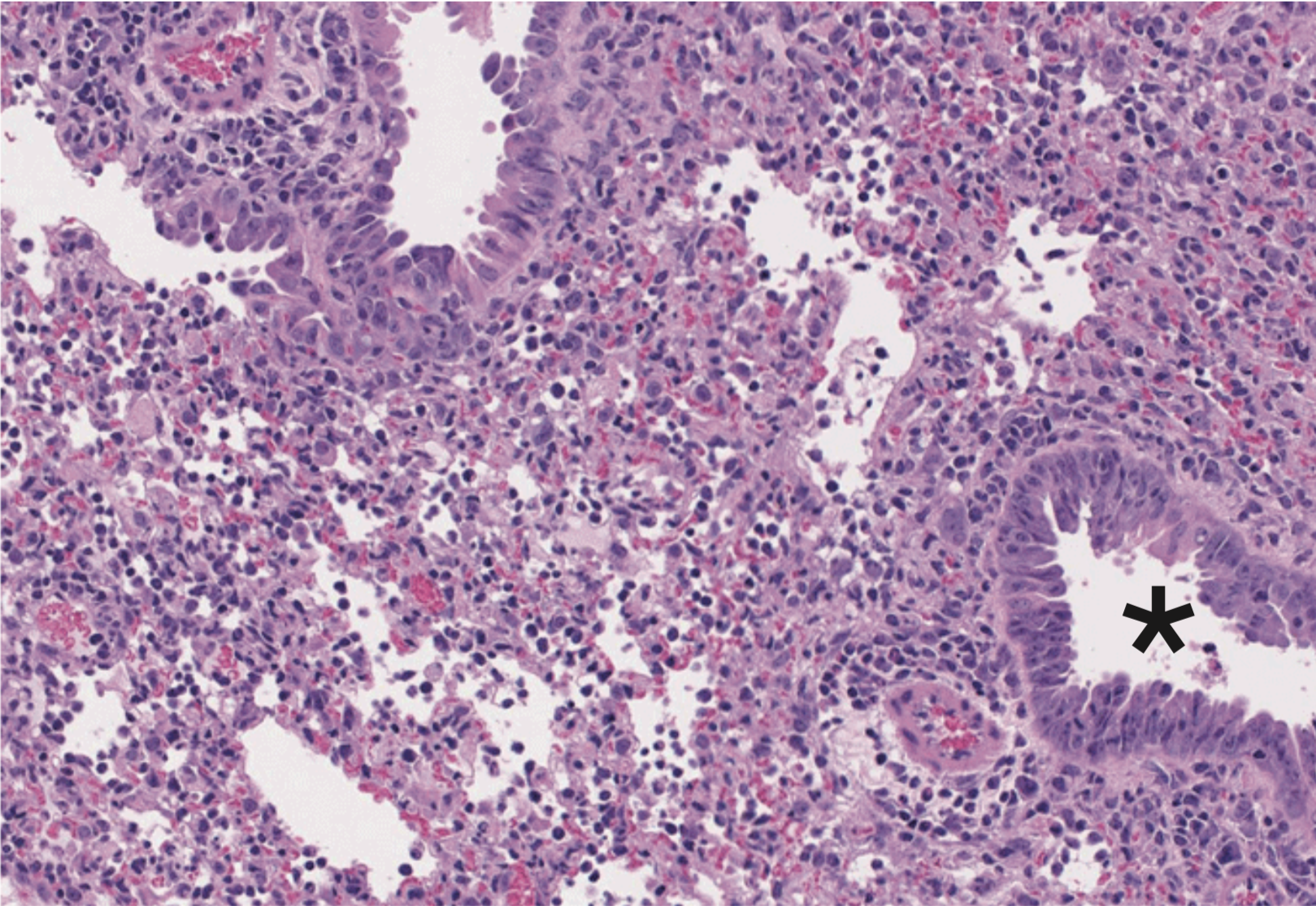
Mucosal barriers serve to interface the internal physiology of an organism with the ever-changing external environment and are colonized by diverse communities of commensal microbes that perform valuable functions to support host metabolism. Constant exposure to novel environmental antigens and high concentrations of microbial ligands increases the risk for persistent inflammatory activation. Accordingly, aberrant immune responses — due to a breakdown in tolerance or defects in barrier maintenance — largely underlie the etiology of chronic mucosal inflammatory disorders. The fine-tuning of opposing immune responses is therefore of even greater relevance at these sites — requiring complex immune networks that operate to contextualize microbial and environmental stimuli. These inputs subsequently shape mucosal immunity and synergize to preserve barrier integrity and function.
